Introduction
Clinical trials generate large amounts of structured and unstructured data, including variables about patient outcomes, investigational drugs, site activity, safety, and regulatory documents.
Robust database management systems enable complete data collection, validation, storage, and analysis while meeting regulatory requirements.
At the same time, clinical trial data have become increasingly valuable beyond operational trial management. Public registries and trial databases now serve as strategic intelligence sources for pharmaceutical companies monitoring:
- drug development pipelines
- emerging therapeutic competitors
- clinical trial site activity
- comparator drug sourcing
- geographic expansion of trials
ClinicalTrials.gov listed 579,013 studies globally as of December 2024, illustrating the vast scope of clinical research data available.
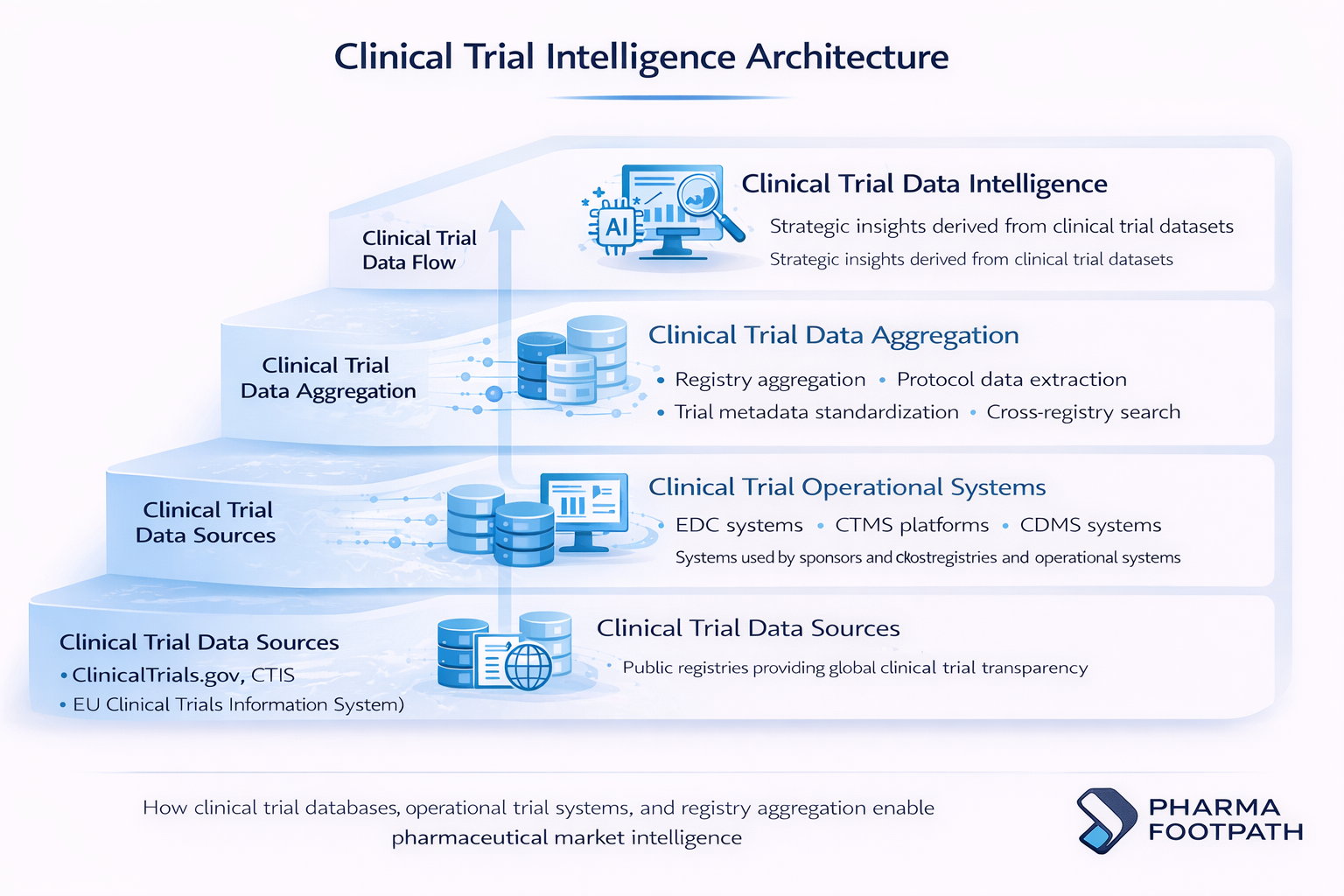
This evolution requires an examination of how internal systems and external registries interact to shape pharmaceutical development.
Next, we define what clinical trial database management entails and why it is essential to the pharmaceutical industry.
In One Sentence
Clinical trial database management is the process of collecting, validating, securing, and analyzing data using systems like EDC, CDMS, or CTMS, while integrating insights from global registries such as ClinicalTrials.gov and CTIS.
What Is Clinical Trial Database Management
Definition
Clinical trial database management refers to the structured handling of data generated during clinical trials, including:
- patient-level clinical data
- investigational product information
- trial protocols and amendments
- investigator and site data
- safety and adverse event reports
The primary objective is to ensure that clinical trial data remains accurate, traceable, and fully compliant with all relevant regulatory standards.
The ICH Good Clinical Practice guideline (ICH E6) establishes international standards for the recording, storage, and reporting of clinical trial data to ensure the integrity and credibility of research results.
Why Clinical Trial Database Management Matters
Effective clinical data management is vital in pharmaceutical R&D.
Regulatory compliance
Regulatory authorities such as:
require sponsors to maintain auditable datasets documenting the entire clinical trial lifecycle.
Data integrity
Clinical decision-making regarding drug safety and efficacy depends on reliable datasets that meet rigorous quality standards.
Transparency
Clinical trial registries ensure public visibility of clinical studies, allowing regulators and researchers to track development programs.
Operational efficiency
Clinical trials are among the most resource-intensive stages of drug development. Effective database management minimizes operational delays and expedites regulatory submissions.

Registry vs Internal Trial Systems
A common confusion is the difference between internal trial systems and public registries.
They serve different purposes.
Public registries complement internal trial systems by enhancing transparency, enabling global trial discovery, and supporting regulatory mandates, rather than replacing them.
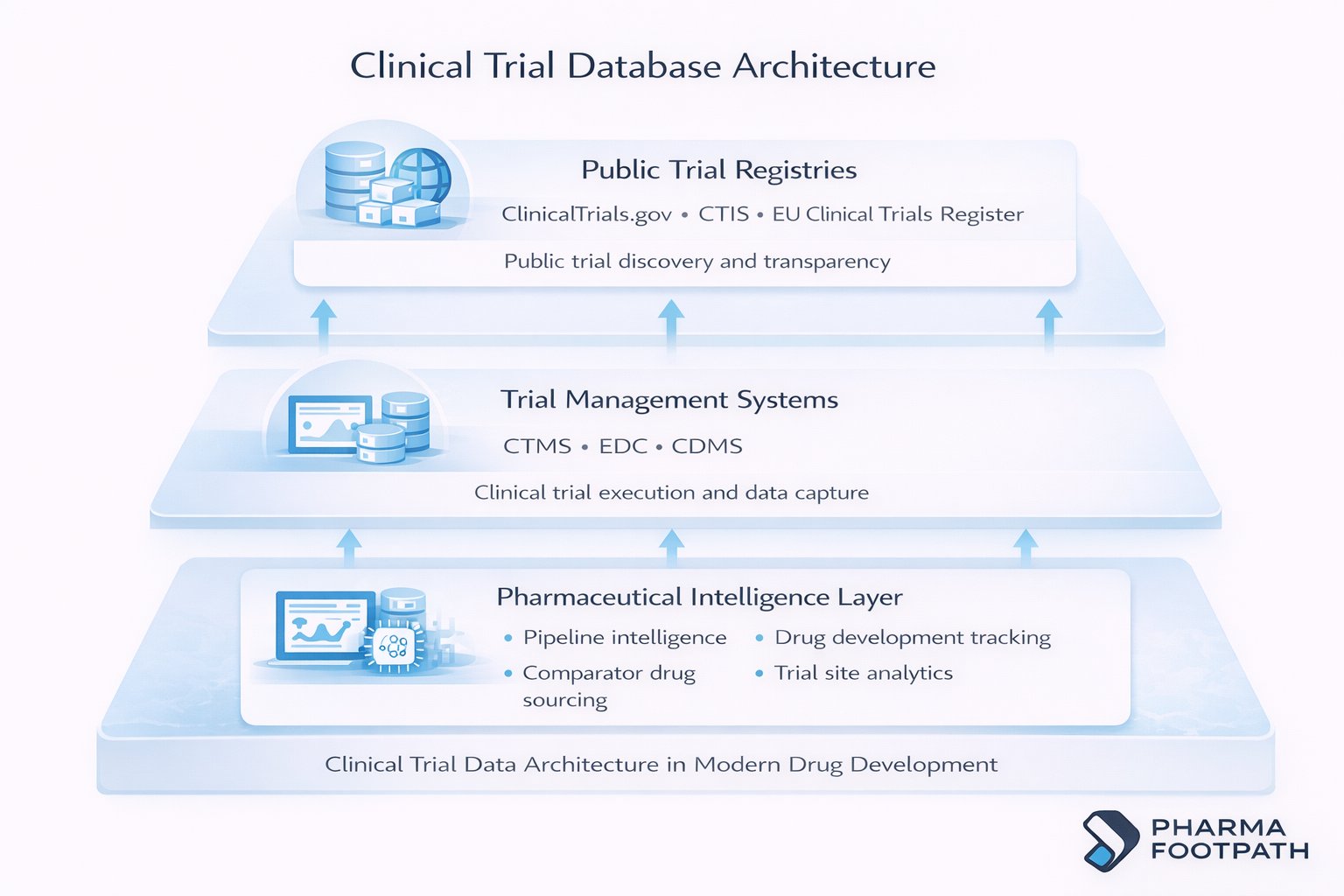
Key Types of Clinical Trial Databases
Several categories of clinical trial database management systems are used.
Clinical Trial Registries
Clinical trial registries are publicly accessible databases that enhance transparency regarding ongoing and completed clinical studies.
Sponsors are required to register specific trials in these systems to satisfy regulatory obligations.
Major registries include:
The WHO International Clinical Trials Registry Platform (ICTRP) acts as a global access point allowing researchers to search across multiple registries simultaneously.
Platforms such as Pharma Footpath help procurement and market intelligence teams analyze clinical trial data alongside pharmaceutical pricing, shortages, and distributor information to identify opportunities faster.
Explore Clinical Trial Intelligence
Clinical Data Management Systems (CDMS)
Clinical Data Management Systems store and validate clinical trial data.
Typical CDMS functions include:
- query management
- dataset validation
- audit trail tracking
- data standardization
CDMS platforms ensure datasets comply with regulatory standards such as:
Electronic Data Capture Systems (EDC)
Electronic Data Capture systems record patient-level data electronically during clinical trials.
Key capabilities include:
- digital case report forms
- automated validation rules
- patient visit tracking
- real-time monitoring.
EDC platforms have largely superseded paper-based data capture methods across the pharmaceutical sector.
Clinical Trial Management Systems (CTMS)
Clinical Trial Management Systems handle operational management of studies.
Typical CTMS functions include:
- site management
- investigator coordination
- budget tracking
- milestone monitoring.
Comparison: CTMS vs EDC vs CDMS vs Public Registries
By combining clinical trial records with pricing, shortages, and product registration data, platforms like Pharma Footpath help teams turn scattered trial information into actionable market insight.
See how Pharmaceutical Market Intelligence works
How Clinical Trial Data Is Collected and Managed
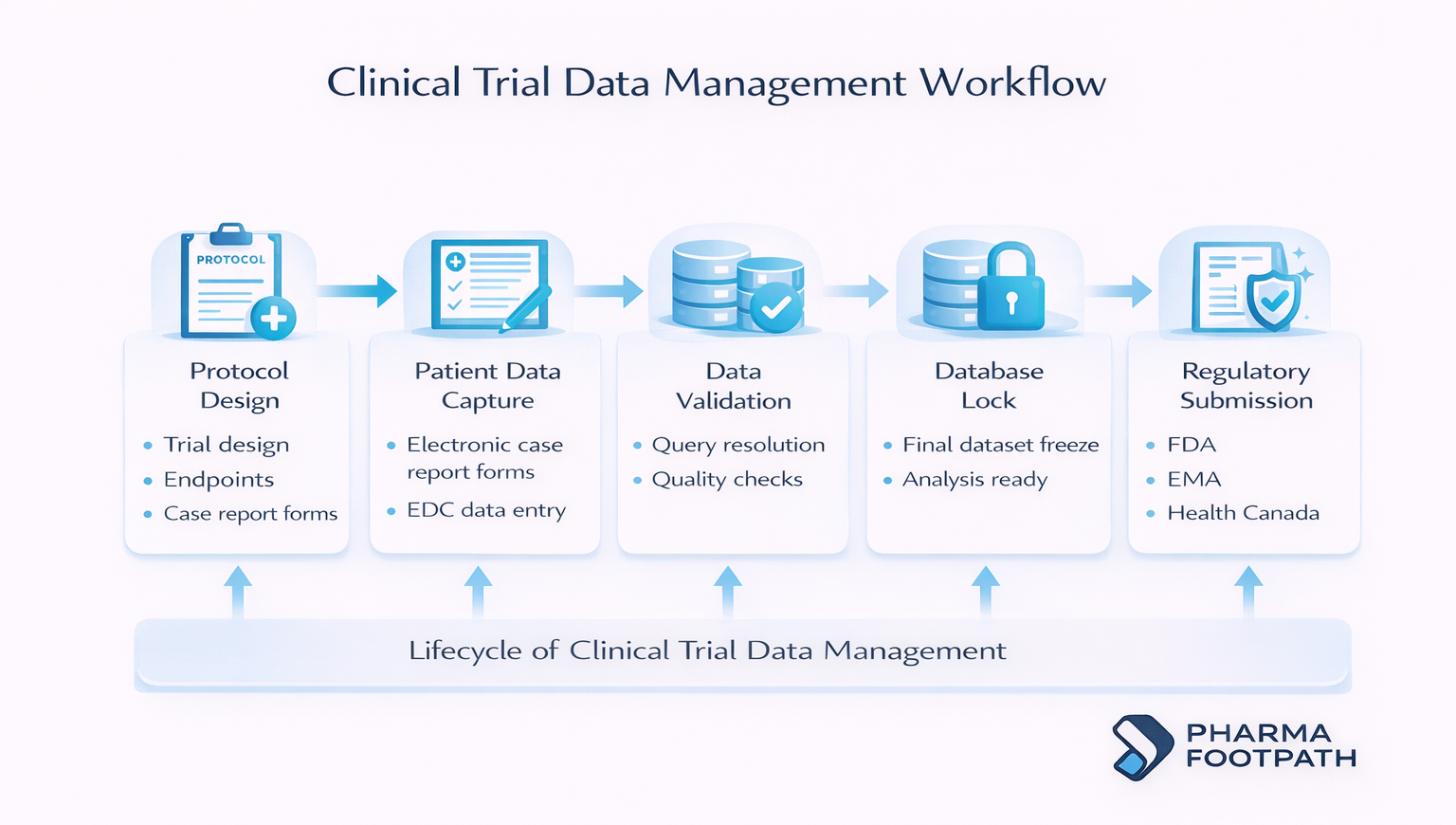
Clinical trial database management follows a structured lifecycle to ensure data integrity and regulatory compliance across all research phases.
Trial protocol setup
Prior to initiating a trial, sponsors are responsible for designing the data collection architecture.
This includes:
- defining endpoints
- designing case report forms
- configuring EDC platforms
Patient data capture
During the trial, investigators systematically collect patient data, including:
- laboratory results
- clinical observations
- imaging data
- patient-reported outcomes.
These data are entered directly into EDC systems to facilitate real-time tracking and validation.
Data validation
Data managers perform quality checks such as:
- missing data queries
- logical consistency checks
- protocol compliance verification.
Database lock
Database lock is the point at which the clinical trial database is finalized, and no further routine changes are permitted before statistical analysis.
This process ensures the dataset prepared for regulatory submissions is both stable and fully validated.
Major Global Clinical Trial Databases
ClinicalTrials.gov
ClinicalTrials.gov is recognized as the world's largest clinical trial registry.
It contains detailed records on:
- study protocols
- recruitment status
- investigators
- trial locations
- results reporting.
As of December 2024, ClinicalTrials.gov listed 579,013 studies worldwide.
Clinical Trials Information System (CTIS)
The Clinical Trials Information System (CTIS) functions as the central European Union platform supporting the Clinical Trials Regulation.
It provides a unified system for:
- clinical trial applications
- regulatory review
- trial oversight
- public transparency.
The CTIS public portal launched on 31 January 2022, allowing researchers and the public to search EU clinical trials.
In April 2025, CTIS became a WHO ICTRP primary registry, strengthening its role in global clinical trial transparency.
EU Clinical Trials Register
The EU Clinical Trials Register provides information on studies conducted under the previous European Union regulatory framework.
While CTIS now serves as the primary EU platform, the register still offers value for historical searches.
Platforms like Pharma Footpath can consolidate these signals and significantly reduce the time required to identify relevant studies, monitor emerging therapies, or analyze competitive pipelines.
For example, centralized intelligence tools can help teams quickly identify trials involving specific active ingredients, therapeutic areas, or geographic markets.
Explore how a Clinical Trial Intelligence Platform can simplify global trial monitoring.
Common Clinical Trial Database Platforms and System Types
Instead of ranking software vendors directly, it is more accurate to categorize the types of systems commonly used in clinical research.
Public registries
- ClinicalTrials.gov
- CTIS
- EU Clinical Trials Register
- Health Canada Clinical Trials Database
- WHO ICTRP
eClinical and internal data platforms
Representative systems include:
- Medidata Rave
- Oracle Clinical One
- Viedoc
- OpenClinica
These platforms support key aspects of clinical trial execution, including data capture, monitoring, and regulatory documentation.
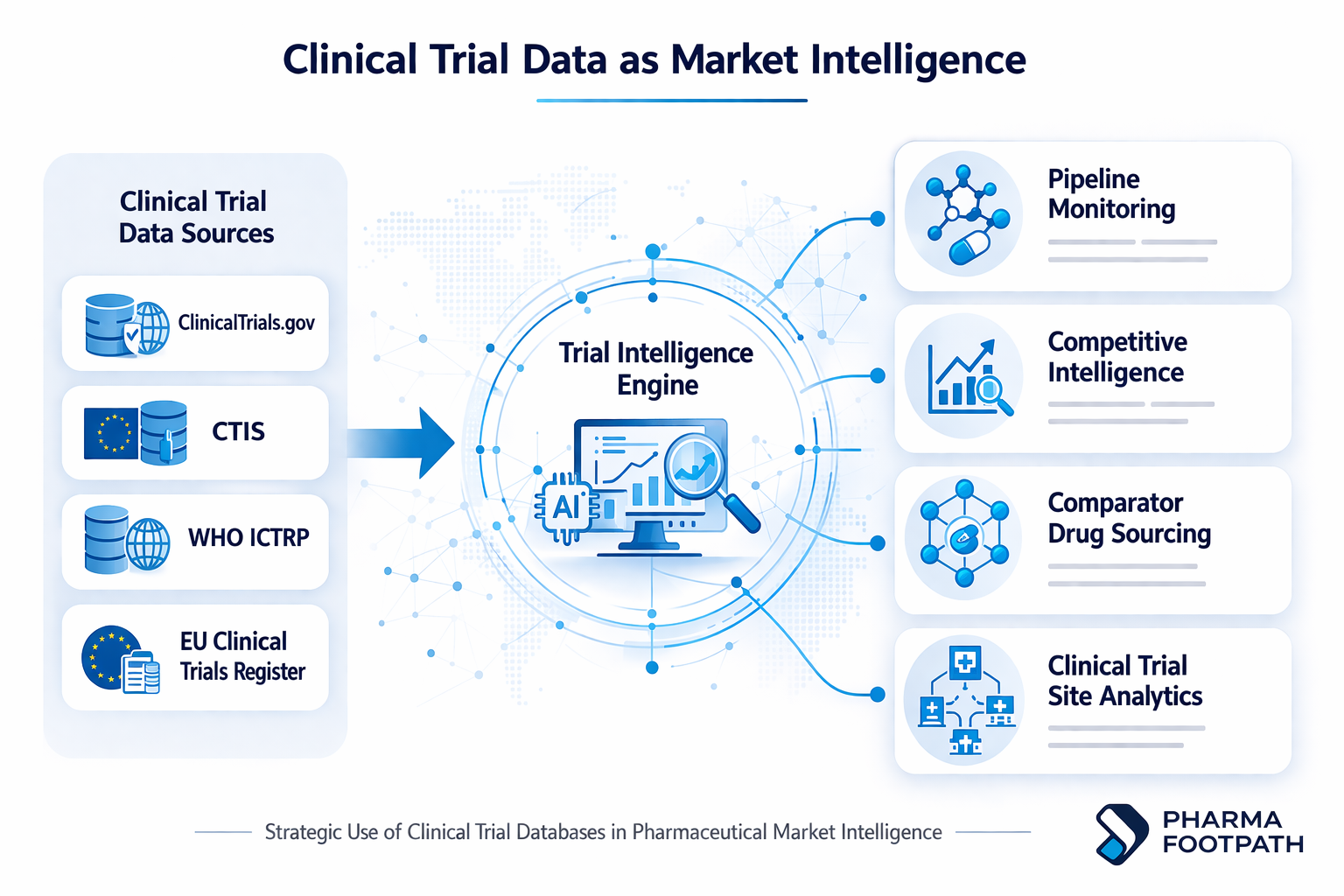
How Pharma Teams Use Clinical Trial Databases
Clinical trial databases increasingly support strategic intelligence workflows.
Pipeline intelligence
Monitoring clinical trial registries helps pharmaceutical companies track:
- investigational compounds
- new therapeutic indications
- competitor development programs.
Competitive intelligence
Clinical trial records provide insights into:
- study sponsors
- comparator drugs
- patient populations
- geographic trial expansion.
These insights help companies anticipate competitor strategies.
Clinical trial sourcing
Clinical trials often require sourcing of comparator drugs, generics, or biosimilars.
Procurement teams may analyze trial databases to identify:
- active ingredients entering trials
- potential supplier markets
- global sourcing opportunities.
Trial feasibility analysis
Trial registries also support decisions about:
- investigator experience
- site recruitment performance
- therapeutic area activity.
When clinical trial records are analyzed alongside pricing data, shortages, and distributor networks, they can provide a much clearer view of the pharmaceutical market intelligence.
Discover Pharma Footpath insights
Best Database for Specific Tasks
Compliance Essentials
Clinical trial database management must align with multiple regulatory frameworks.
Key compliance pillars include:
- ClinicalTrials.gov registration requirements under U.S. federal law
- EU Clinical Trials Regulation transparency rules via CTIS
- ICH E6(R3) guidance on computerized systems and data security
ICH E6(R3) emphasizes validated computerized systems, oversight of the data lifecycle, and the secure management of digital clinical trial records.
Clinical Trial Database Management Challenges
Despite technological advances, several structural challenges remain.
Fragmented data sources: Clinical trial information is distributed across many registries and systems globally.
Data standardization: Different registries and trial platforms use varying data formats.
Regulatory complexity: Sponsors must comply with regulatory frameworks across multiple jurisdictions.
Future Trends in Clinical Trial Data Management
Several trends are reshaping clinical trial data management.
AI-driven trial analytics: Artificial intelligence is increasingly used to analyze large clinical trial datasets.
Global clinical trial intelligence platforms: Integrated platforms now aggregate trial registries and other pharmaceutical datasets.
Real-time trial monitoring: Digital trial platforms enable sponsors to continuously monitor recruitment and trial performance.
Why Clinical Trial Databases Matter for Procurement and Market Intelligence
Clinical trial data is increasingly relevant beyond research operations.
For procurement and supply-chain teams, trial databases can reveal:
- emerging biosimilar programs
- upcoming generic competition
- investigational molecules entering late-stage trials
- geographic trial expansion patterns.
Public trial registries are useful, but they often need to be combined with pricing, regulatory, and supply chain intelligence to support commercial decisions.
Platforms that aggregate these signals enable pharmaceutical teams to link trial activity to broader market dynamics.
Pharma Footprint brings these datasets together, helping pharmaceutical companies monitor clinical trials while analyzing drug prices, shortages, and distributor networks across global markets.
FAQ
What is a clinical trial database?
A clinical trial database is a structured system storing information about clinical studies, including protocols, investigators, trial sites, and results.
What is clinical trial database management?
Clinical trial database management refers to the processes for capturing, validating, securing, and analyzing clinical trial data throughout the study lifecycle.
What is the difference between CTMS and EDC?
CTMS manages operational aspects of clinical trials such as site management and milestones, while EDC captures patient-level clinical data in electronic case report forms.
What is CTIS?
CTIS is the Clinical Trials Information System used across the EU/EEA for submission, assessment, and oversight of clinical trials under the Clinical Trials Regulation.
What is ClinicalTrials.gov?
ClinicalTrials.gov is a public registry and results database of clinical research studies. It listed over 579,000 studies worldwide as of December 2024.
What is database lock in clinical trials?
Database lock is the stage at which a clinical trial dataset is finalized, allowing statistical analysis to begin.
Is CTIS the same as the EU Clinical Trials Register?
No. CTIS is the newer system introduced under the EU Clinical Trials Regulation, while the EU Clinical Trials Register is the earlier registry used under the previous regulatory framework.
Conclusion
Clinical trial database management has evolved from a purely operational task into a strategic capability for pharmaceutical organizations.
Modern clinical research relies on multiple data infrastructures, including:
- Electronic Data Capture platforms
- Clinical Trial Management Systems
- Clinical Data Management Systems
- global trial registries such as ClinicalTrials.gov and CTIS.
Together, these systems support the capture, validation, and analysis of clinical trial data while ensuring regulatory transparency.
For pharmaceutical companies, however, the value of clinical trial data extends beyond trial execution. Registries increasingly serve as early indicators of drug development activity, competitive pipelines, and sourcing opportunities.
Public registries provide important transparency, but their insights are most powerful when combined with broader pharmaceutical intelligence.
By integrating clinical trial data with pricing intelligence, product registrations, shortages, and distributor networks, platforms such as Pharma Footpath enable pharmaceutical procurement teams and market analysts to monitor drug development trends and identify sourcing opportunities across global markets.
About the Author

Phil Thomas has had a varied background, including tech product management and international trade of pharmaceuticals. While primarily in clinical trial supply, he supported companies in unlicensed medicines, parallel trade, drug shortages, and managed access programs.
He has previously sold a big data business and served as managing director at a cybersecurity consultancy. He has a large collection of unread books, which he promises to read one day.






