Top 10 Pharmaceutical Wholesalers in the UK
Updated 5/12/26
Introduction to the Pharmaceutical Market in the UK
The UK pharmaceutical supply chain relies on a small number of major wholesale distributors, supported by specialist and short-line wholesalers.
These companies ensure medicines reach pharmacies, hospitals, dispensing doctors, and clinical trial sites while upholding regulatory and cold-chain standards.
Understanding the market’s scale and composition is essential.
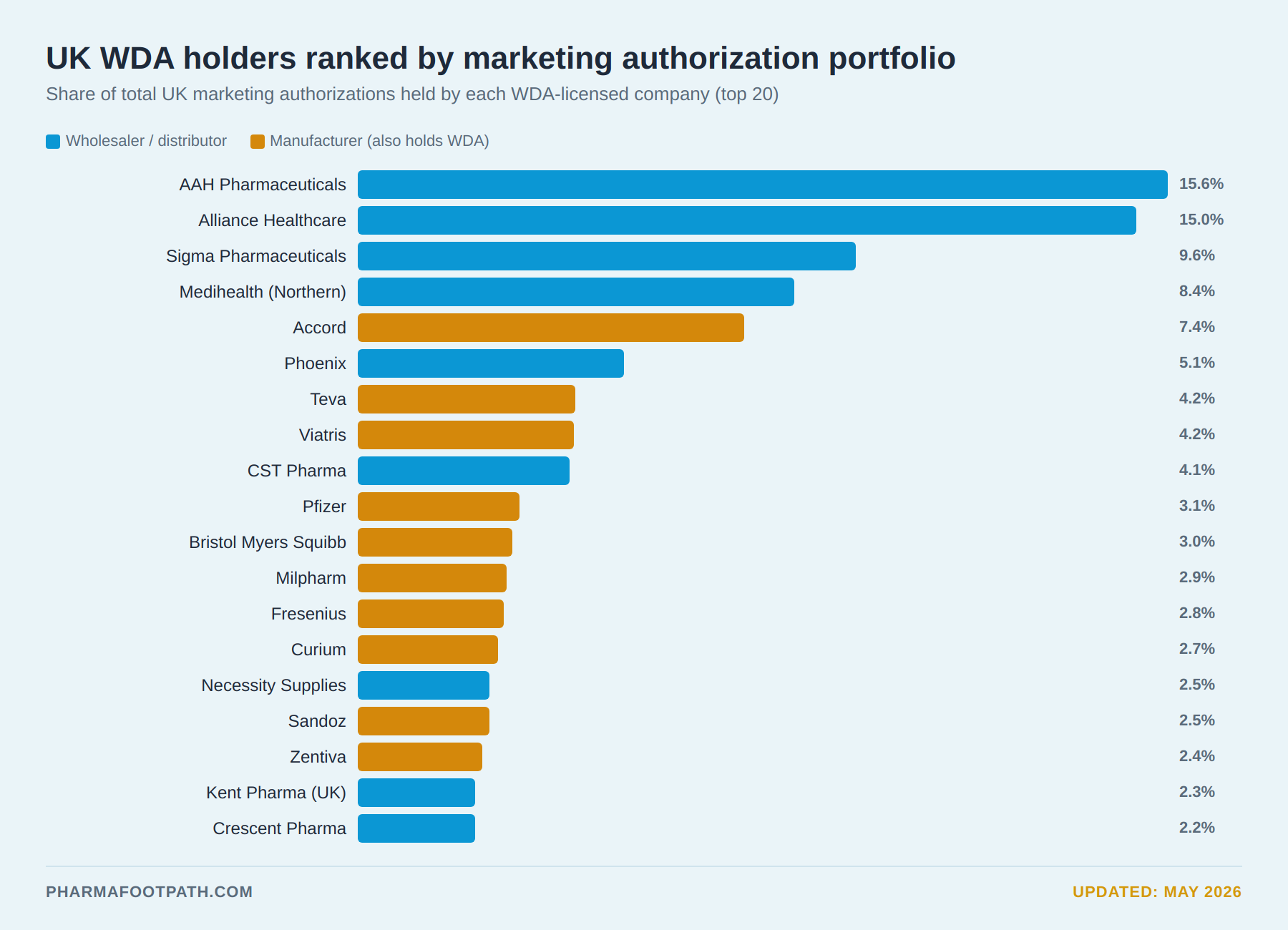
The UK pharmaceutical wholesaling sector, projected to be worth £68.5 billion in 2026 and comprising approximately 2,878 businesses, is dominated by major players, including Alliance.
The Market Problem
Ongoing medicine shortages, parallel trade, and supply chain disruptions place greater pressure on procurement teams to maintain a stable pharmaceutical supply.
Procurement professionals should move beyond basic sourcing and conduct a thorough, criteria-based evaluation of each supplier’s ability to meet organizational needs. Key factors to consider include:
- MHRA licensing status
- Good Distribution Practice (GDP) compliance
- cold-chain capability
- NHS supply experience
- specialty medicine access
- shortage-response capacity
- cross-border sourcing capabilities
As pharmaceutical supply chains become more fragmented, procurement teams increasingly rely on pharmaceutical market intelligence platforms such as Pharma Footpath to compare distributors, monitor shortages, review product registrations, and identify sourcing opportunities across the UK and European markets.
This guide helps procurement teams navigate the complex landscape by reviewing the top UK pharmaceutical wholesalers, explaining sector operations, and outlining effective comparison methods.
This approach enables readers to evaluate options based on the challenges discussed above.
What Is a Pharmaceutical Wholesaler?
A pharmaceutical wholesaler purchases medicines from manufacturers and distributes them to pharmacies, hospitals, healthcare providers, dispensing doctors, and other authorized buyers.
Wholesalers act as the operational bridge between pharmaceutical manufacturers and healthcare systems. Their responsibilities include:
- medicine storage and warehousing
- inventory management
- GDP-compliant transportation
- cold-chain handling
- product traceability
- controlled drug procedures
- emergency and shortage-response supply
The UK pharmaceutical wholesale market includes both full-line wholesalers and specialist short-line distributors.
Pharmaceutical Wholesaler vs Distributor
The terms “wholesaler” and “distributor” are often used interchangeably, but they can refer to slightly different operational models.
A pharmaceutical wholesaler typically purchases and resells medicines to healthcare customers. A distributor may operate broader logistics services on behalf of manufacturers, including warehousing, pre-wholesale operations, and direct-to-pharmacy delivery.
In practice, many large UK companies perform both functions.

Full-Line vs Short-Line Wholesalers
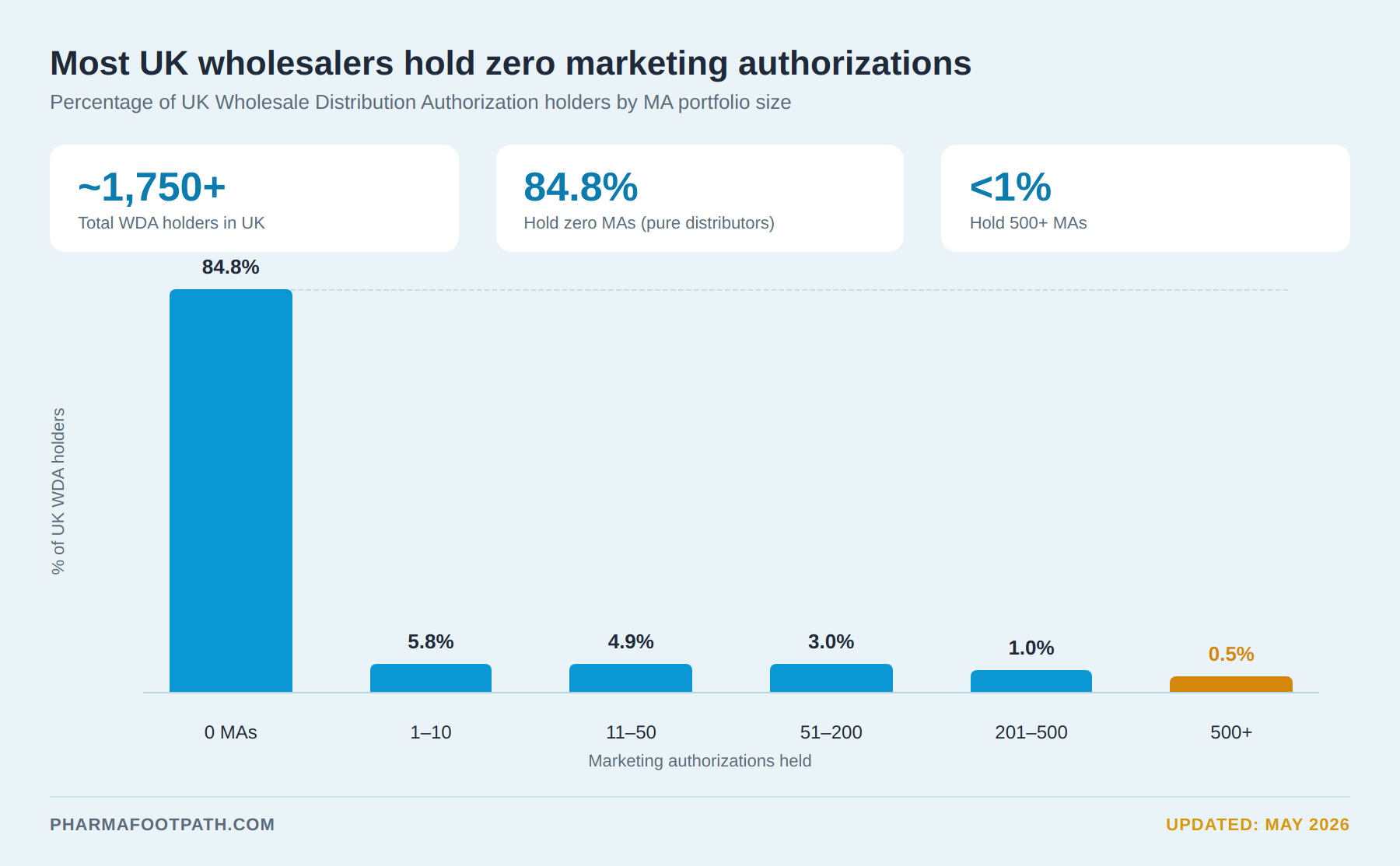
The British Association of Pharmaceutical Wholesalers previously noted that although the UK had around 1,700 wholesale dealer licenses, only a limited number of true full-line wholesalers operated nationally.
Why MHRA Licensing Matters
All UK wholesale dealers handling human medicines must hold a valid MHRA Wholesale Distribution Authorization (WDA(H).
This authorization confirms that the company complies with UK Good Distribution Practice (GDP) requirements covering:
- medicine storage
- transportation
- traceability
- recall procedures
- temperature control
- quality systems
For procurement teams, verifying WDA(H) status is a basic yet essential supplier validation step.
How We Ranked the Top UK Pharmaceutical Wholesalers
Since many UK pharmaceutical wholesalers are privately held and do not disclose full financial data, our ranking uses a transparent, multi-dimensional set of criteria.
These criteria reflect operational performance and market impact, focusing on attributes relevant to procurement teams. Each wholesaler was assessed consistently across these factors to ensure transparency and fairness, rather than relying solely on revenue.
Instead, the companies below were evaluated using:
- UK market presence
- NHS relevance
- pharmacy and hospital coverage
- cold-chain and GDP capability
- specialist sourcing capability
- distribution infrastructure
- clinical trial supply capability
- public licensing and operational visibility
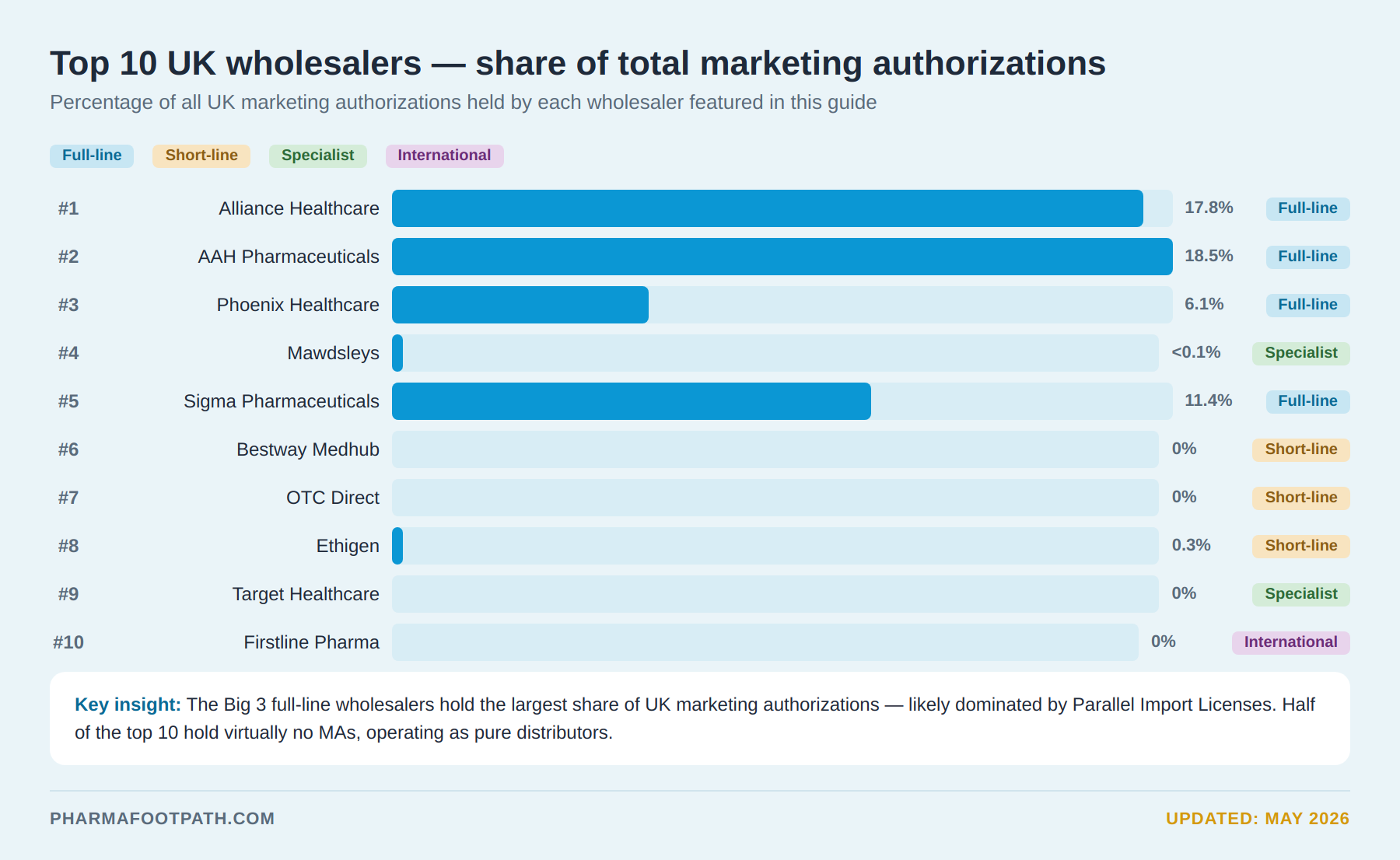
Top 10 Pharmaceutical Wholesalers in the UK
1. Alliance Healthcare
Alliance Healthcare, part of global company Cencora, is a key UK and European distributor supporting broad pharmacy and healthcare networks.
Alliance Healthcare delivers medicines to over 115,000 care points in Europe, leveraging extensive logistics and product coverage.
Its scale, combined with its significant NHS involvement, evidenced by its participation in NHS framework agreements as an approved supplier, establishes Alliance Healthcare as a leading UK wholesaler.
2. AAH Pharmaceuticals
AAH Pharmaceuticals is a top full-line UK wholesaler, serving pharmacies, hospitals, and dispensing doctors nationwide.
AAH Pharmaceuticals is deeply integrated into the UK medicines distribution system, with a national infrastructure and product range essential to reliable supply, especially within NHS-linked systems.
AAH Pharmaceuticals’ national infrastructure and broad product portfolio enable reliable access to medicines across the UK.
Its regional distribution centers provide daily, temperature-controlled deliveries to rural pharmacies, reducing the risk of stockouts during periods of high demand.
This operational capacity supports consistent patient care, minimizes supply disruptions, and optimizes inventory management, key objectives for pharmacy procurement.
3. Phoenix Healthcare Distribution
Phoenix Healthcare Distribution is part of the PHOENIX Group, one of Europe’s largest pharmaceutical wholesalers.
PHOENIX Group spans 29 markets with more than 210 distribution sites.
Phoenix Healthcare Distribution specializes in the distribution of UK medicines, pharmacy services, and pharmaceutical care. The company maintains strong relationships with the NHS and community pharmacies throughout the UK.
4. Mawdsleys
Mawdsleys is a leading UK independent pharmaceutical wholesaler.
Unlike some traditional full-line wholesalers, Mawdsleys has built strong capabilities in:
- hospital medicines
- clinical trial supply
- pharmaceutical logistics
- specialty medicines
- international supply-chain services
Mawdsleys’ ability to manage clinical trial logistics and distribute specialty medicines supports research and complex programs in UK hospitals.
5. Sigma Pharmaceuticals
Sigma Pharmaceuticals is an established wholesaler serving independent UK pharmacies and providers.
The company provides pharmacy supplies, product availability, and distribution support for community pharmacies.
6. Bestway Medhub
Bestway Medhub operates in the pharmacy supply space, with a strength in generics, OTC products, and procurement support.
Bestway Medhub appeals to pharmacies seeking alternatives and competitive prices.
7. OTC Direct
OTC Direct operates as a short-line wholesaler specializing in:
- generics
- parallel imports
- surgical products
- OTC medicines
Short-line wholesalers like OTC Direct are vital during shortages or price changes.
8. Ethigen
Ethigen supplies generic medicines and products to UK pharmacies and providers.
The company focuses on generic and value pharmaceutical wholesale.
9. Target Healthcare
Target Healthcare supports pharmacy and specialist sourcing, including the procurement of niche medicines.
Specialist wholesalers step in when full-line distributors face shortages.
10. Firstline Pharma
Firstline Pharma focuses on specialist sourcing, import/export, and supply-chain support.
International sourcing capability is crucial as procurement teams seek alternative supply routes during shortages.
For a broader European market overview, see our article on the Largest Pharmaceutical Wholesalers in Europe, which analyzes major distributors such as PHOENIX Group, Alliance Healthcare, McKesson Europe, and other leading pharmaceutical wholesale networks operating across EU markets.
Who Are the Top 3 Pharmaceutical Wholesalers in the UK?
The UK wholesale market is generally dominated by three major full-line distributors:
- Alliance Healthcare
- AAH Pharmaceuticals
- Phoenix Healthcare Distribution
IBISWorld identifies Alliance Healthcare, AAH Pharmaceuticals, and Phoenix Healthcare Distribution among the largest companies operating in the UK pharmaceutical wholesaling market.
These wholesalers operate nationwide and supply pharmacies, hospitals, and healthcare providers.
Which UK Pharmaceutical Wholesalers Supply the NHS?
NHS procurement operates through framework agreements and approved supplier structures.
Public NHS procurement documentation identifies suppliers, including:
- AAH Pharmaceuticals
- Alliance Healthcare
- Mawdsley-Brooks
- Phoenix Healthcare Distribution
These companies support the supply of medicine for the NHS and healthcare sector purchasing operations.
NHS-facing wholesalers must demonstrate strong regulatory compliance, reliable delivery, adherence to GDP standards, and the capability to plan for contingencies.
How to Check if a UK Pharmaceutical Wholesaler Is Licensed
Use the MHRA Wholesale Distribution Register
The Medicines and Healthcare products Regulatory Agency maintains a public register of licensed wholesale distribution sites.
Procurement teams should verify:
- active WDA(H) authorisation
- company site details
- licensed activities
- scope of operations
Check WDA(H), Site Address, and Product Scope
A wholesaler’s authorization may apply only to specific sites or operational activities.
Always confirm:
- exact operating entity
- warehouse location
- authorised activities
- product categories handled
Verify GDP and Cold-Chain Capabilities
For biologics, vaccines, specialty medicines, and clinical trial products, procurement teams should also assess:
- GDP compliance procedures
- temperature-controlled logistics
- cold-chain monitoring
- recall procedures
- controlled drug handling
UK Pharmaceutical Wholesalers and Medicine Shortages
Medicine shortages have become a major operational issue across both the UK and EU healthcare systems.
The European Medicines Agency has stated that shortages place significant pressure on healthcare systems and create risks for patient care.
Research supported by EMA and the Pharmaceutical Group of the European Union found that European pharmacists spent an average of 10.6 hours per week managing medicine shortages in 2024.
The UK government and the Department of Health and Social Care have taken active steps to strengthen medicine supply management and shortage-response planning, as demonstrated by the introduction of the 2022 Medicines Supply Contingency Planning Program and the updated Serious Shortage Protocols (SSPs), which provide legal frameworks for managing and mitigating supply disruptions across the NHS.
For procurement teams, shortages increase the importance of:
- alternative supplier identification
- parallel import visibility
- cross-border sourcing intelligence
- distributor comparison
- stock-risk monitoring
Procurement Scenario
A hospital pharmacy sourcing a critical injectable medicine may encounter:
- allocation limits from a primary wholesaler
- cold-chain restrictions
- delayed manufacturer deliveries
- parallel trade disruption
In these situations, procurement teams require immediate access to comprehensive supplier information in order to make time-sensitive, strategically significant decisions.
Rapid visibility is essential for assessing alternative sourcing options, mitigating supply interruptions, and maintaining continuity of patient care amid risk and uncertainty.
- alternative licensed wholesalers
- EU sourcing options
- product registrations
- availability across multiple markets
- specialist distributors with GDP-compliant cold-chain capability
During medicine shortages, procurement teams often need to identify alternative distributors quickly while assessing licensing status, geographic coverage, and product availability.
Platforms such as Pharma Footpath aggregate distributor intelligence, shortage data, product registrations, and pricing signals to help sourcing teams evaluate supply options more efficiently across the UK and EU markets.

How Procurement Teams Should Compare Pharmaceutical Wholesalers
Procurement decisions should extend beyond pricing alone.
Key comparison criteria include:
How Pharma Footpath Helps Identify Pharmaceutical Wholesalers
With supply chains becoming more fragmented and shortages more frequent, procurement teams need dynamic, evidence-based distributor intelligence rather than static supplier lists.
Continuously updated intelligence provides detailed insights into supplier performance, regulatory compliance, shortage response, and real-time availability.
This approach enables procurement professionals to anticipate disruptions, rigorously evaluate suppliers, and make data-driven decisions to mitigate supply risks and maintain continuity of care.
Pharma Footpath helps sourcing and procurement teams:
- identify pharmaceutical distributors
- compare medicine pricing across markets
- monitor shortages
- analyse product registrations
- track parallel trade activity
- evaluate sourcing opportunities
- support clinical trial sourcing intelligence
This type of visibility becomes particularly valuable when procurement teams need to quickly identify alternative suppliers during supply disruptions.
FAQ
Who is the largest pharmaceutical distributor in the UK?
Alliance Healthcare, AAH Pharmaceuticals, and Phoenix Healthcare Distribution are generally considered among the largest pharmaceutical distributors in the UK. IBISWorld identifies these companies as major industry players.
What is a pharmaceutical wholesaler?
A pharmaceutical wholesaler buys, stores, and distributes medicines to pharmacies, hospitals, dispensing doctors, and healthcare providers while complying with regulatory and GDP standards.
What is the difference between a full-line and short-line wholesaler?
A full-line wholesaler supplies a broad range of medicines nationally, while a short-line wholesaler focuses on selected products such as generics, parallel imports, or OTC medicines.
Do UK pharmaceutical wholesalers need an MHRA license?
Yes. UK pharmaceutical wholesalers handling human medicines must hold a valid MHRA Wholesale Distribution Authorization (WDA(H).
Which wholesalers supply the NHS?
NHS framework suppliers include AAH Pharmaceuticals, Alliance Healthcare, Mawdsley-Brooks, and Phoenix Healthcare Distribution.
Why are pharmaceutical wholesalers important?
Wholesalers ensure medicines move efficiently from manufacturers to pharmacies, hospitals, and healthcare providers while maintaining traceability, storage, and regulatory compliance.
How can procurement teams compare UK wholesalers?
Procurement teams should compare licensing status, GDP compliance, product availability, cold-chain capability, NHS experience, geographic coverage, shortage-response capability, and EU sourcing access.
How can I find reliable pharmaceutical suppliers in the UK?
Reliable supplier identification should include verification of MHRA licensing, GDP compliance, distribution capability, shortage performance, and sourcing coverage across the UK and EU markets.
Conclusion
The UK pharmaceutical wholesale market is led by a relatively small number of major full-line distributors supported by specialist wholesalers, short-line suppliers, and international sourcing partners.
In summary, procurement teams should move beyond supplier lists and adopt a systematic evaluation of pharmaceutical wholesalers.
We recommend actively assessing each wholesaler’s licensing, distribution infrastructure, shortage management, pricing transparency, cold-chain capability, and cross-border sourcing.
Ongoing supply chain disruptions highlight the need for advanced distributor intelligence platforms, such as Pharma Footpath, to support data-driven decisions and risk mitigation.
Integrating these criteria will strengthen the availability and resilience of medical services, supporting both patient care and organizational goals.
About the Author

Phil Thomas has had a varied background, including tech product management and international trade of pharmaceuticals. While primarily in clinical trial supply, he supported companies in unlicensed medicines, parallel trade, drug shortages, and managed access programs.
He has previously sold a big data business and served as managing director at a cybersecurity consultancy. He has a large collection of unread books, which he promises to read one day.










