Introduction
Pharmaceutical pricing varies significantly between national markets, even within highly integrated regulatory regions such as the European Union. A medicine reimbursed at a lower price in Spain or Greece may cost substantially more in Germany, Sweden, or Denmark due to differences in national pricing policies, reimbursement negotiations, and procurement systems.
Due to price differences, businesses have a commercial opportunity to move goods from lower-priced markets to higher-priced markets - this process is known as pharmaceutical parallel import.
Parallel imports allow authorised medicines to be purchased in one EU or EEA country and supplied in another through independent distributors rather than the manufacturer’s official distribution network. Although the product remains the same medicine, the distribution channel differs.
For procurement teams, distributors, and supply chain professionals, understanding how parallel imports work is essential for navigating pharmaceutical pricing differences, sourcing medicines across markets, and responding to supply disruptions.
Key Takeaways
- Pharmaceutical parallel import refers to the legal practice of purchasing medicines in one EU country and selling them in another through independent distributors outside the manufacturer’s official supply network.
- Parallel trade is driven by price differences between national pharmaceutical markets, which arise from different government pricing and reimbursement policies.
- In the European Union, parallel imports account for roughly €6–7 billion in annual sales, representing approximately 2.6–2.8% of the total pharmaceutical market.
- Parallel imported medicines must be essentially identical to the reference medicinal product already authorised in the destination country and must receive regulatory approval before distribution.
- Some countries rely heavily on parallel trade. For example, parallel imports account for up to 25% of pharmacy medicine sales in Denmark, highlighting their role in certain national supply chains.
- For procurement teams, parallel imports provide alternative sourcing channels, enabling organizations to respond to price differences and temporary supply shortages.
- As pharmaceutical supply chains become increasingly complex, market intelligence tools that track pricing, product registrations, and distributor networks are becoming essential for identifying parallel trade opportunities.
What Is Pharmaceutical Parallel Import?
Definition of Parallel Import
Pharmaceutical parallel import refers to the practice of purchasing an authorised medicinal product in one country and importing it into another for resale without the original manufacturer’s authorisation.
In the European Union, this typically involves a distributor acquiring a medicine from a wholesaler in one member state where prices are lower and supplying the same product to another member state where prices are higher.
The defining feature of parallel import is that the medicine is produced by the original manufacturer but distributed through an independent supply channel rather than the manufacturer’s official distribution network.
Parallel import is legal within the EU under the principle of intellectual property exhaustion and the free movement of goods.
Parallel Import vs Parallel Distribution
Parallel import refers to medicines authorised through national marketing authorisation procedures. In this case, a distributor obtains a parallel-import license from the destination country's national medicines authority.
Parallel distribution applies to medicines authorised through the European Medicines Agency's centralised procedure. These medicines receive a single EU-wide authorisation granted by the European Commission.
The distinction is regulatory rather than commercial. In both cases, medicines are distributed across borders outside the manufacturer’s official supply network while remaining authorised products.
Key Differences Between Parallel Trade Models
Why Parallel Imports Exist in the Pharmaceutical Industry
Medicine Price Differences Between Countries
The primary economic driver of parallel imports is price variation across national pharmaceutical markets.
Unlike many consumer products, pharmaceutical prices are often regulated at the national level. Governments negotiate drug prices with manufacturers or impose reimbursement frameworks that determine the prices healthcare systems pay.
When a medicine is cheaper in one country than another, distributors may take advantage by purchasing in the lower-priced market to supply the higher-priced one.
This practice, commonly known as pharmaceutical price arbitrage, underpins parallel trade.
EU Single Market Rules
Parallel imports are also enabled by the legal framework of the European single market.
The EU principle of free movement of goods allows products legally marketed in one member state to circulate across other member states, provided public health protections are maintained.
Pharmaceutical parallel trade operates within the broader legal framework governing intra-European commerce.
Once legally marketed in one EU country, medicines may be resold in other EU countries by independent distributors, provided they have regulatory approval.
Intellectual Property Exhaustion
Parallel imports are also supported by the principle of regional exhaustion of intellectual property rights within the European Economic Area. Once a manufacturer places a product on the market in the EEA, its distribution rights for that specific product are considered exhausted.
This allows independent distributors to resell medicines across EU markets, provided regulatory requirements are met.
Monitoring these pricing differences across multiple markets requires structured pharmaceutical market data. Platforms such as Pharma Footpath help procurement teams analyze pricing, licensing, and distributor information across EU pharmaceutical markets.
How Pharmaceutical Parallel Import Works
The process of parallel importing medicines typically follows several stages.
The first stage involves identifying price differences between national pharmaceutical markets. Parallel trade companies monitor medicine prices across EU countries to find opportunities to source the same product at a lower cost in another market.
Once a sourcing opportunity is found, the importer buys the medicine from wholesalers or authorised suppliers in the exporting country. The importer then applies for a parallel import licence from the medicines authority in the destination country.
Regulators evaluate the application to confirm that the imported medicine is essentially identical to the locally authorised reference medicinal product. Only after regulatory approval can the importer begin supplying the product within the destination market.
In many cases, the medicine must also be repackaged or relabeled to meet national language and packaging requirements. After these adjustments, the product can be distributed to pharmacies, hospitals, and healthcare providers through pharmaceutical wholesalers.
Parallel Trade in the European Pharmaceutical Market
Parallel trade represents a relatively small but influential segment of the European pharmaceutical supply chain.
Industry analysis indicates that parallel imports account for approximately €6–7 billion in annual sales across Europe, equivalent to around 2.6–2.8% of total pharmaceutical spending.
Although the overall market share is modest, its impact varies across countries. In several Northern European markets, including Denmark, Sweden, and the Netherlands, parallel imports account for a much larger share of the pharmacy supply.
Germany remains the largest parallel import market in Europe, with estimated annual sales of approximately €2.9 billion.
For procurement teams analyzing these dynamics, access to structured market data is essential.
Platforms such as Pharma Footpath aggregate pharmaceutical pricing, regulatory approvals, and distributor information to help companies better understand cross-border medicine trade patterns.
The infographic below summarizes the key dynamics of pharmaceutical parallel trade in Europe, including market size, supply chain flows, and sourcing patterns across countries.

Understanding parallel trade requires combining pricing data, regulatory information, and distributor networks across multiple markets.
Platforms such as Pharma Footpath help procurement teams analyze these factors and identify sourcing opportunities across EU pharmaceutical markets.
Regulatory Framework for Parallel Imports
Pharmaceutical parallel imports operate within a regulated environment designed to ensure patient safety.
Each EU member state has a national medicines authority that reviews parallel import applications. These authorities verify product equivalence, manufacturing standards, and pharmacovigilance before granting a license.
For stakeholders monitoring cross-border distribution, understanding how to identify parallel trade licenses in the EU is essential for tracking product movement and regulatory activity.
Agencies such as Germany’s Federal Institute for Drugs and Medical Devices or the Belgian Federal Agency for Medicines confirm that the imported product matches the reference medicinal product already authorised in the market.
Parallel import regulation also interacts with the broader European pharmaceutical regulatory framework, including Directive 2001/83/EC, which governs medicinal products for human use.
Parallel Trade Licensing Landscape in Europe
Across the European pharmaceutical market, parallel trade operates through a large and fragmented licensing framework.
According to Pharma Footpath research, Industry estimates indicate that there are more than 95,000 parallel trade licenses across the EU, of which roughly 75,000 are national-level parallel import licenses, while the remainder relate to centrally authorised medicines.
This distinction is important for market intelligence. Parallel distribution approvals for centrally authorised medicines are publicly visible through European Medicines Agency notifications, whereas national-level parallel import licenses are issued by individual regulatory authorities and are not consolidated in a single EU-wide database.
As a result, monitoring parallel trade activity across Europe often requires collecting data from multiple national regulatory sources rather than relying on a single centralized register.
Benefits of Parallel Imports
Parallel imports can play several roles within pharmaceutical markets.
One key benefit of parallel imports is increased price competition. When parallel importers supply medicines to higher-priced markets, additional suppliers compete with the original manufacturer’s network. This competition can reduce medicine prices and create cost savings for healthcare systems.
Evidence from European markets illustrates this effect. For instance, a 2017 analysis by the Danish Ministry of Health reported that parallel imports led to annual savings of approximately 700 million Danish kroner.
Parallel trade can also improve supply resilience. When shortages occur in one market, distributors may source medicines from other EU countries where supply is more abundant.
Challenges and Risks
Despite its benefits, parallel trade also raises operational challenges. One frequently discussed concern is the potential for supply imbalances between countries when large volumes of medicines are exported from lower-priced markets.
Parallel trade also involves complex regulatory compliance requirements, including licensing procedures, packaging rules, and pharmacovigilance obligations across jurisdictions.
Real-World Examples of Parallel Trade
Parallel imports are widely used in European pharmaceutical supply chains.
In oncology markets, where specialty medicines can have substantial price differences across countries, procurement teams often explore cross-border sourcing options. Parallel trade companies specialize in identifying price differentials and facilitating imports.
Parallel Trade Source Markets Vary by Country
Parallel import flows are not uniform across Europe. Different destination markets tend to source medicines from specific exporting countries depending on price differences, reimbursement policies, and wholesaler networks.
For example, analysis of parallel trade licenses shows that Italy and Poland source parallel imports from distinct sets of origin markets, reflecting regional price structures and established distribution channels.
Market dynamics can vary significantly across countries, as illustrated by the Italian parallel import market, where data fragmentation complicates analysis.
The following infographics illustrate the leading countries of origin supplying parallel-imported medicines to these markets.
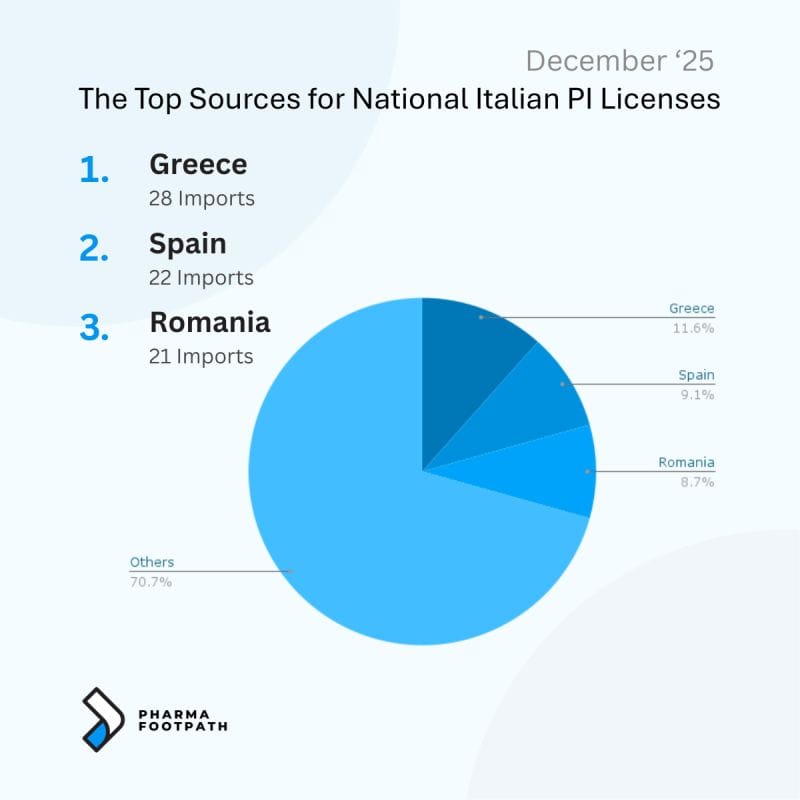
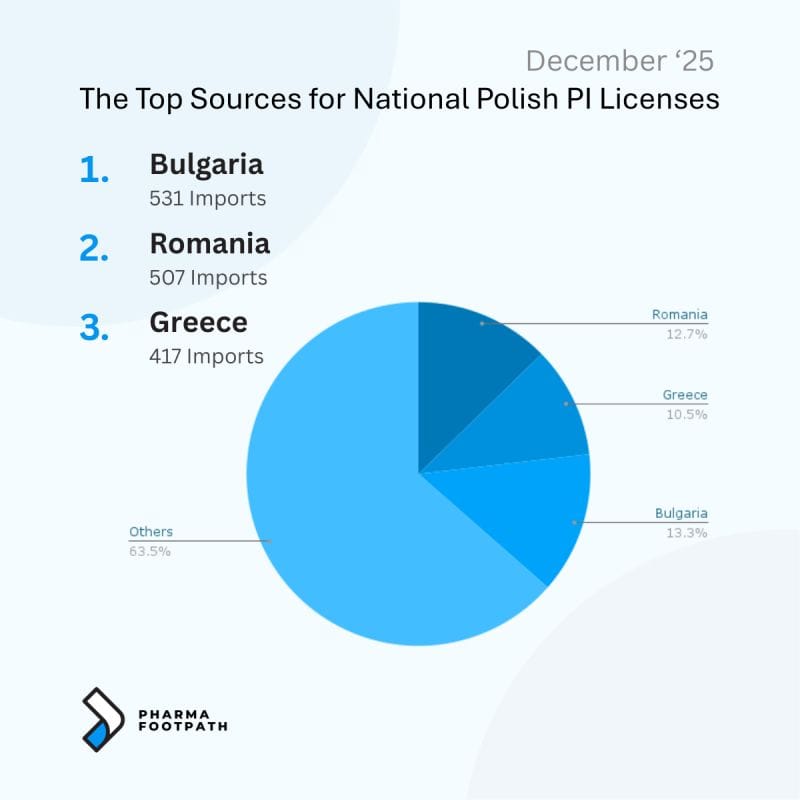
How the Pharmaceutical Parallel Import Supply Chain Works
Pharmaceutical parallel imports involve a structured supply chain linking manufacturers, distributors, regulators, and healthcare buyers across multiple markets.
Independent distributors monitor price differences across countries, source medicines from lower-priced markets, obtain regulatory approval in the destination market, and distribute the products through wholesalers to pharmacies and hospitals.
The process generally unfolds in several stages.
Parallel Import Process
Parallel trade is possible because of the EU principle of free movement of goods and the exhaustion of intellectual property rights, which allow medicines legally marketed in one EU country to be resold in another under regulated conditions.
Procurement Strategy: Identifying Parallel Import Opportunities
Identifying viable parallel trade opportunities requires monitoring pharmaceutical market data across multiple countries.
Procurement teams typically analyze signals such as price differences between markets, distributor availability, regulatory approvals, and medicine shortage alerts.
Historically, gathering this information required manual monitoring of national regulatory databases, wholesaler networks, and market reports.
As pharmaceutical supply chains become more complex, companies increasingly rely on pharmaceutical market intelligence platforms to consolidate data from these sources.
Platforms such as Pharma Footpath aggregate information on medicine pricing, product registrations, distributor networks, and regulatory licenses.
Procurement teams increasingly rely on a pharmaceutical parallel trade intelligence platform to consolidate pricing, licensing, and distributor data across EU markets.
The Future of Pharmaceutical Parallel Trade
Drug shortages, pricing transparency, and increasing supply chain complexity are likely to influence the role of parallel imports in the coming years.
As pharmaceutical markets become more interconnected, procurement teams increasingly rely on accurate and timely market intelligence to monitor cross-border trade and identify sourcing opportunities.
FAQ
What is parallel import in pharma?
Parallel import in pharmaceuticals refers to purchasing a medicine in one country and importing it into another country for resale outside the manufacturer’s official distribution network.
Is pharmaceutical parallel import legal?
Yes. Parallel imports are legal within the European Union because they operate under the principle of free movement of goods within the EU single market.
Are parallel-imported medicines safe?
Yes. Parallel imported medicines must be essentially identical to the reference product already authorised in the importing country and must receive regulatory approval before being marketed.
What is the difference between parallel import and parallel distribution?
Parallel import applies to nationally authorised medicines, whereas parallel distribution applies to medicines authorised centrally by the European Medicines Agency.
Why do pharmaceutical prices vary between countries?
Medicine prices vary due to national reimbursement systems, government negotiations with pharmaceutical companies, and differences in healthcare policy between countries.
Track Pharmaceutical Parallel Trade Opportunities with Market Intelligence
Parallel trade opportunities can emerge quickly when price differences shift between markets or when supply disruptions affect certain countries. However, identifying these opportunities requires continuous monitoring of regulatory licenses, distributor networks, and pharmaceutical pricing data across Europe.
Pharma Footpath’s Pharmaceutical Parallel Trade Intelligence solution helps procurement teams and distributors analyze:
- medicine pricing differences across markets
- parallel trade licenses and regulatory approvals
- distributor and wholesaler networks
- product registrations and supply signals
By consolidating these datasets, pharmaceutical organizations can identify sourcing opportunities earlier and gain clearer visibility into cross-border medicine trade.
Speak with our team about pharmaceutical parallel trade intelligence
Platforms that aggregate pharmaceutical trade intelligence enable companies to make more informed sourcing decisions in a market characterized by price differentials, regulatory complexity, and supply chain disruptions.
Conclusion
By enabling authorised medicines to move across borders outside manufacturer-controlled distribution channels, parallel trade introduces additional competition and provides procurement teams with alternative sourcing options.
However, navigating parallel trade requires visibility into pricing differences, regulatory approvals, distributor networks, and supply dynamics across multiple countries.
Market intelligence platforms such as Pharma Footpath help pharmaceutical organisations monitor these signals by aggregating data on medicine pricing, parallel trade licenses, distributor activity, and product registrations.
Access to reliable pharmaceutical market intelligence enables procurement teams to better understand cross-border trade in medicines and identify potential sourcing opportunities in an increasingly complex supply environment.
About the Author

Phil Thomas has had a varied background, including tech product management and international trade of pharmaceuticals. While primarily in clinical trial supply, he supported companies in unlicensed medicines, parallel trade, drug shortages, and managed access programmes.
He has previously sold a big data business and served as managing director at a cybersecurity consultancy. He has a large collection of unread books, which he promises to read one day.







