
February may have been a shorter month, but we've managed to pack in some significant improvements!
Latest updates
New markets - USA and Switzerland


Two new important markets for PharmaFootpath! USA is obviously a global leader in pharmaceutical products, with the latest treatments being launched here. There's been some interesting challenges with adding this data, both with the way that we capture it, translate it to our standards, and then just the sheer volume of it! It's been great to have overcome these issues, and include this data.
With regards to pricing data, we're capture pricing around the prices paid by pharmacies and medicine organisations AFTER discounts have been applied, rather than list price. This real world data helps for companies looking to launch or supply products into the US, but can be radically different from the price companies looking to import US products from exporters.
New sign-in process
We've added Single Sign On to significantly improve the security posture of our applicaiton, following a review from a cybersecurity consultant. You will have received an invite to the new process if you're an active user, as old login credentials will no longer work. There's still some improvements to make on this, with an improvement to the appearance of the login screen as an example, so you may see some further small changes as we make some further final adjustments
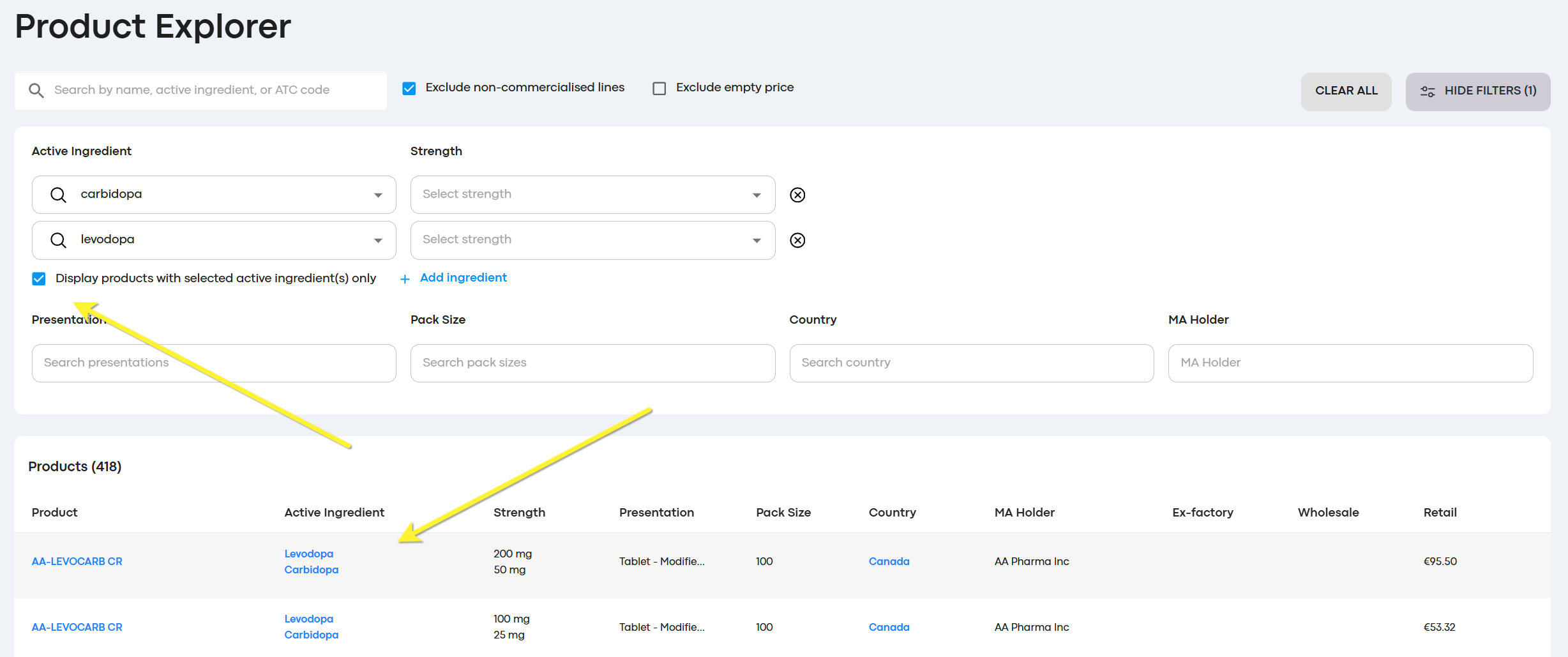
Exact searching in active ingredients
We received user feedback that the advanced search was useful, but returning results for all products for an active ingredient, including those products where it was one of a combination was less than ideal. We've added an option to filter results to the exact match. This could be one active ingredient, or multiple like in the example below(e.g. show me the products where Levodopa and Cabridopa are the active ingredients AND there are no further active ingredients)
Newsletter
We've been posting Linkedin updates on the latest EMA changes to product registrations, but after some user feedback we've turned this into a newsletter also to make it easier to share. In addition, we've also started to add in information about changes to shortages at the EMA level, though these aren't very frequent.
If you'd like to see other information added in to this, please let me know and I'll see what we can include!
Minor improvements
- strengths alligned with active ingredients in products with multiple active ingredients
- UK(Northern Ireland) removed from EU filter (and USA, which accidentally snuck in there when we added this market somehow!)
- Security enhancements and controls
- Minor visual appearance improvements in tables
- Adding sorting by origin in the parallel trade table
- Other very small adjustments
March plans
Automation of more markets - This continues to be our main priority, and will likely be for the next 3 months.
Saying goodbye to the ATC code tree - Our observations are this isn't well used, and we'd prefer to trim out things unnecesary features to focus on the things that add most value
2 factor authentication - We'll be adding in mandatory 2fa for all users, which will provide significant security improvements
Looking ahead later to later this year, we'll be attending Expopharm in Düsseldorf, September 16-18th. This will be the first time that we'll have a stand at a conference, and I hope we can book in plenty of meetings with you in advance!
All the best,
Phil



